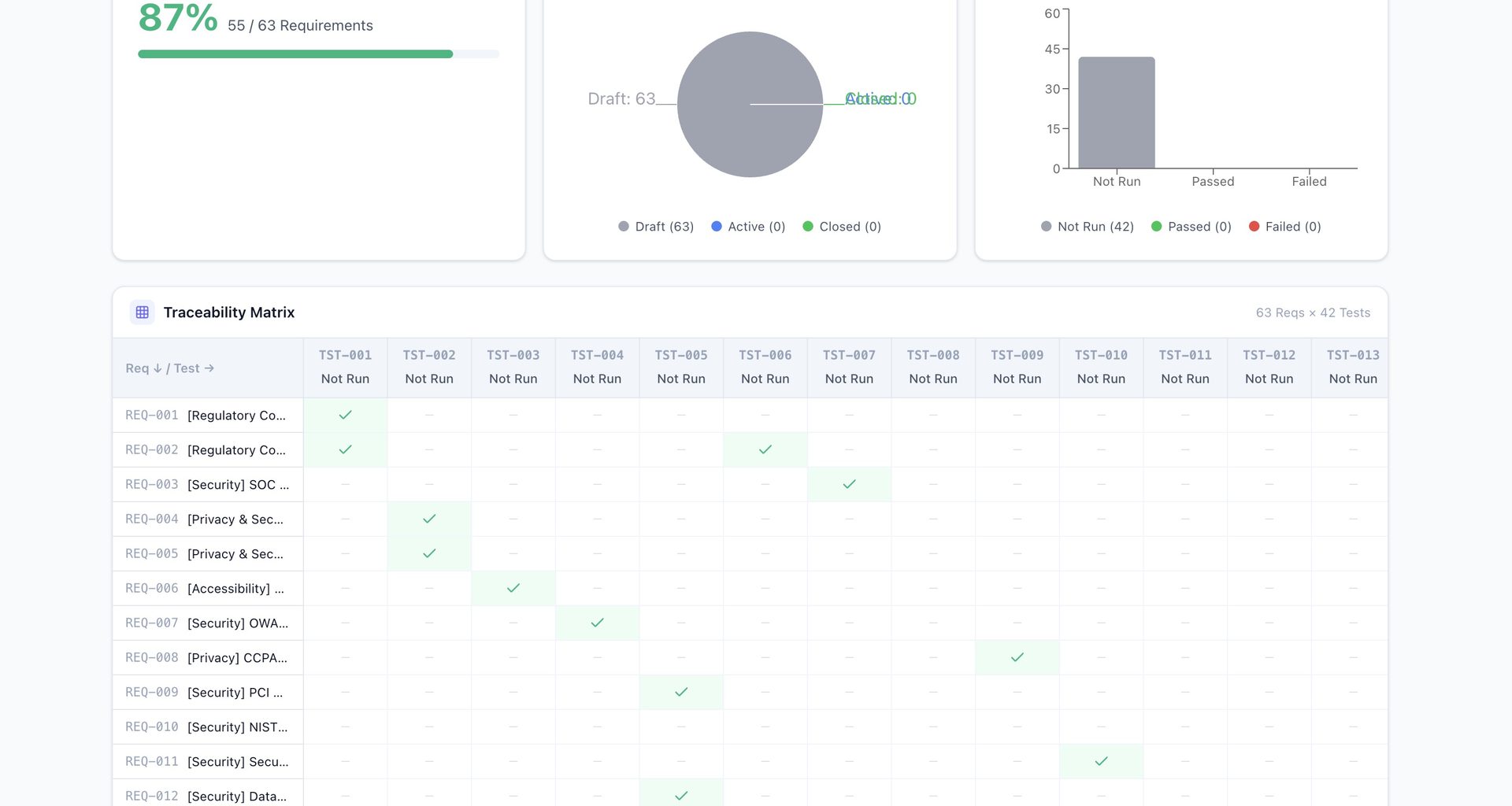
Market Context
QMS Software Market: $12.5B (2025) → $31.5B by 2034 (10.81% CAGR)

The FDA Computer Software Assurance Implementation Manual: Risk-Based Assurance Case Frameworks, Quality System Software Documentation, Test Protocol … Device & Life-Science Quality Operations
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Revenue Impact by Release
| Release | Target Customers | Est. Market Opportunity |
|---|---|---|
| v3.0 QMSR/Device (shipped) | ~6,500 FDA device companies + ~30,000 EU MDR manufacturers | QMSR transition creates $500M+ tooling demand |
| v3.1 Enterprise | Multi-site pharma/biotech (top 200 + CMOs) | Enterprise QMS: $4B segment |
| v3.2 Clinical/AI | ~1,100 CROs globally + pharma sponsors | eTMF market alone: $2B+ |
| v3.3 Global | Emerging market manufacturers | Brazil, India, Saudi, UAE: $1.5B+ QMS spend |
ISO 13485 gap assessment tool
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
v3.0 Features — Why They Matter NOW
FDA QMSR (effective Feb 2, 2026)
- Replaces decades-old 21 CFR 820 QSR with ISO 13485:2016 incorporation by reference
- Every FDA-regulated device company must demonstrate ISO 13485 conformity
- FDA stopped using QSIT inspection technique — now risk-based inspections
- QAtrial’s ISO 13485 Gap Assessment directly addresses this transition
Device Recall Crisis
- 115% increase in device recalls since 2018
- $5B+/year cost to industry
- #1 source of FDA 483 observations: Design Control failures
- QAtrial’s Design Control Kanban directly addresses this
EU AI Act (deadline Aug 2, 2027)
- High-risk AI medical devices must document: data governance, bias mitigation, algorithm transparency
- MDCG 2025-6 confirms AI Act requirements can integrate into ISO 13485 QMS
- QAtrial’s AI provenance tracking + gap analysis supports this
![Express Schedule Free Employee Scheduling Software [PC/Mac Download]](https://m.media-amazon.com/images/I/41yvuCFIVfS._SL500_.jpg)
Express Schedule Free Employee Scheduling Software [PC/Mac Download]
Simple shift planning via an easy drag & drop interface
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Customer Demand Signals
| Pain Point | Priority | QAtrial Feature |
|---|---|---|
| Cross-functional collaboration silos | #1 pain point | Workflow Engine, Notifications |
| Rising regulatory scrutiny (QMSR, MDR, AI Act) | Accelerating | ISO 13485 Assessment, Gap Analysis |
| Legacy system limitations (Part 11, audit trail) | Persistent | Audit Trail, E-Signatures, RBAC |
| Data volume overwhelming manual processes | Growing | AI Co-Pilot (test gen, risk, gap, CAPA) |
| Operational demands without headcount increase | Universal | AI automation, template composition |
| Vendor lock-in with expensive tools | $10K-100K+/user/year | Open-source AGPL-3.0, self-hosted |
AI-powered QMS for medical devices
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Competitive Landscape
| Capability | QAtrial (Free) | Greenlight Guru ($25K+/yr) | MasterControl ($50K+/yr) | Veeva Vault ($100K+/yr) |
|---|---|---|---|---|
| ISO 13485 Gap Assessment | ✅ Static + AI | ❌ | Partial | Partial |
| Design Control (DHF/DMR/DHR) | ✅ | ✅ | ✅ | ✅ |
| AI Test Generation | ✅ Bring your own LLM | ❌ | ❌ | ❌ |
| AI Risk Classification | ✅ Multi-taxonomy | ❌ | ❌ | ❌ |
| QMSR Transition Tool | ✅ 27-clause mapping | ❌ | ❌ | ❌ |
| Self-hosted / Air-gapped | ✅ | ❌ | ❌ | ❌ |
| Source Code Auditable | ✅ AGPL-3.0 | ❌ Proprietary | ❌ Proprietary | ❌ Proprietary |
| Multi-language (12+) | ✅ | ❌ | Partial | ✅ |
| Country × Vertical Templates | ✅ 37 × 10 | Device-only | Generic | Pharma-focused |
| Approval Workflows | ✅ Configurable | Basic | ✅ | ✅ |
| Cost | $0 | ~$25K/yr | ~$50K/yr | ~$100K+/yr |
Key Takeaway
QAtrial v3.0 shipped at the exact moment the market needs it most — FDA QMSR goes live in 2026, device recalls are at historic highs, and companies are looking for alternatives to $50K-100K/year quality tools. The combination of open-source transparency + AI co-pilot + ISO 13485 gap assessment is something no commercial competitor offers.